|
Update: An edited version of this article has been published by Antibody Genie. Thanks to Dr. Colm Ryan for the invitation. Check out the article here: www.antibodygenie.com/blog/can-infection-cause-chronic-disease/
With non-communicable diseases (or NCDs, which include heart disease, diabetes, cancer) now responsible for the most deaths worldwide, large investments into research on these diseases are helping us understand their causes. Many of these diseases have something in common, they involve chronic inflammation. Cells normally triggered by the immune system to fight infection, are persistently activated by factors produced within the body (endogenous factors), eventually causing damage to bodily tissues and beginning the disease processes involved in the development of diabetes, cancer and atherosclerosis - the build-up of fatty plaques in the arteries causing heart attacks and strokes. This begs the question, if both infection and endogenous factors can trigger the same immune responses that are known to cause NCDs, could infection directly cause NCDs?
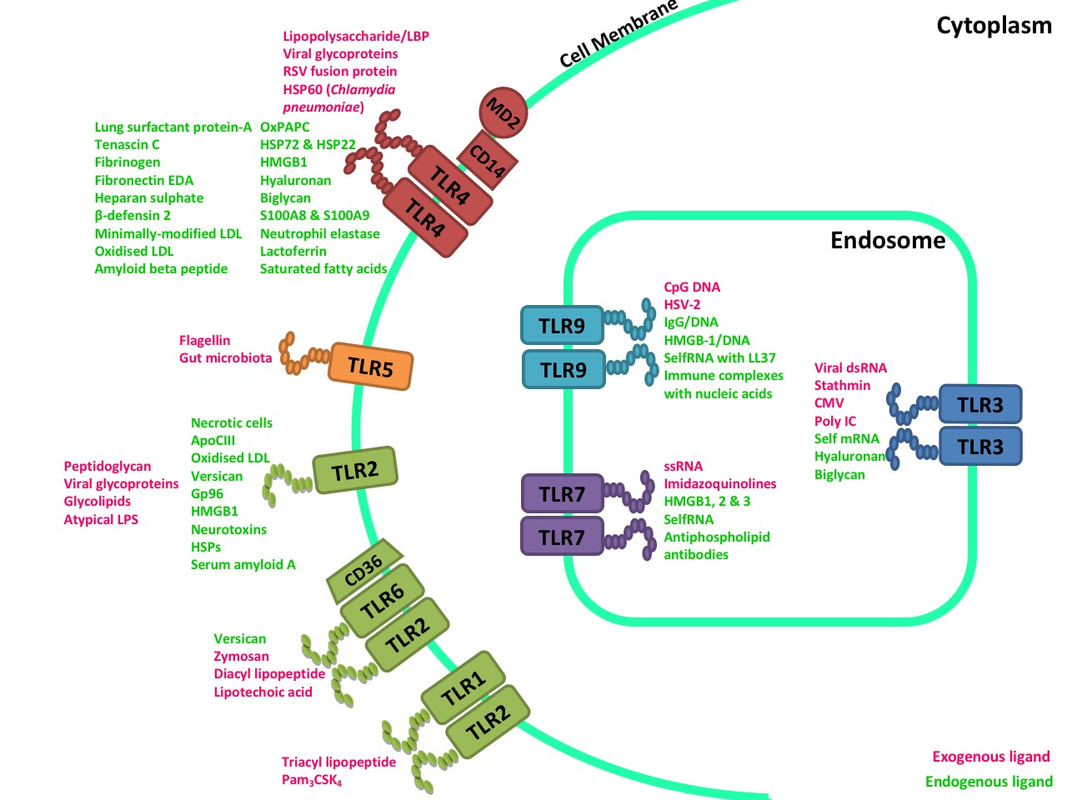
Triggering our immune defences A good example of the responses triggered by both infection and endogenous factors starts with the well-characterised family of Toll-like receptors (TLRs), which are found on the surface of many immune cells. These receptors play a key role in innate immunity – the first line of defence against infection. Each of the 9 members of the TLR family recognise different components of pathogens, for example TLR4 recognises lipopolysaccharide (LPS) found on the outer membrane of certain types of bacteria, and TLRs 3 and 7 recognise viral ribonucleic acids or RNAs, which viruses release into the host cell they infect to exploit the cell’s machinery to produce its damaging viral proteins. When TLRs bind to a molecule they recognise, they can activate a sequence of proteins ending with a protein called NFkB (NFkappaB) and/or the Interferon regulatory factor (IRF) family, which are responsible for the production of numerous inflammatory proteins [1]. Once an immune response is triggered following TLR activation, it is usually sufficient to successfully destroy the infection. Thus, the infection is short-lived and the immune system can return to a resting state.
There are numerous examples of endogenous factors produced in the body, that can also activate TLRs, and they can be produced continuously over a long period of time, unlike the usually brief insult caused by infection. Therefore, endogenous factors are usually behind the development of chronic disease. For example, free fatty acids derived from a diet with high levels of fat, are known to activate TLRs. High blood glucose levels can activate TLRs and increase levels of free fatty acids in the blood, thus increasing the risk of diabetes and obesity. It is well known that in atherosclerosis, TLRs recognise numerous endogenous factors. For example oxidised low density lipoprotein (oxLDL) and saturated fatty acids, from high fat meals, can be recognised by TLR2 and TLR4 [2]. Crucially TLRs are found on the surface of the cell that is the main culprit in worsening atherosclerotic disease - the macrophage. When TLRs become active on a macrophage for a prolonged period, resulting in increased production of inflammatory proteins, macrophages become less effective in clearing harmful oxLDL, ultimately dying and depositing themselves in the artery wall leading to atherosclerotic plaque build-up [3]. Hence it is plausible to believe that chronic activation of TLRs by serious, lingering infections could increase the incidence of heart attacks, strokes, diabetes and other NCDs.
The known links between infections and NCDs
A direct link has been proven between viral infection and diabetes. Viruses can use the art of ‘molecular mimicry’ where they mimic proteins found within our bodies and direct our immune systems to attack our own body tissues. This occurs when TLRs found on the surface of dendritic cells, recognise viruses and erroneously direct T cells to target our own tissues. As a result these T cells damage islet cells in the pancreas (which produce insulin and other hormones), eventually causing diabetes [5]. Recent studies have revealed that infections significantly increase the likelihood of atherosclerosis and subsequent heart disease, and an aggregation of several infections increases the burden further [6]. Infections that have been associated with heart disease include the hepatitis A virus, herpes simplex virus, Chlamydia pneumoniae and influenza. Amazingly live influenza virus has been recovered from atherosclerotic arteries even after the illness had resolved, further supporting a contribution of viral infection to atherosclerotic disease [7]. Viral DNA has also been recovered from atherosclerotic plaques [8]. While a direct link between infection, immune activation and atherosclerosis has yet to be established, more and more studies are suggesting that such a hypothesis is not unreasonable. All TLRs found on the surface of cells, and many of the proteins they subsequently activate have been shown to increase inflammation and worsen atherosclerosis [4]. One example of such a protein is IRF5, which I recently showed prevents the clearance of dead cells in the artery, thus worsening plaque build-up [9], which you can read more about here. So it may only be a matter of time before a direct link is established between infection and atherosclerosis. Why should we study the link between infection and NCDs? We typically think of diseases like heart disease and obesity as being most prevalent among mature, sedentary, obese individuals in the developed world, but the reality is very different. NCDs are now more prevalent in developing countries and the patients are getting younger, hence NCDs are primarily diseases of poverty [10]. Could it be that people in developing countries are at greater risk of contracting infections, due to poorer hygiene and healthcare, hence the prevalence of NCDs is also increasing? Infections are not the only reason inflammation occurs in our bodies, other factors such as smoking, stress and poor diet play a role, and may also be a greater problem in developing countries. But it is not unreasonable to think that tackling infectious diseases could ease the burden of disease on a population in more ways than one. Our fight against serious communicable diseases such as HIV/AIDS and Malaria could also indirectly reduce the incidence of NCDs. Thus, further research into the links between infections and NCDs could significantly improve quality of life and life expectancy in the developing world.
References
[1] Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010 ;11(5):373–384. www.nature.com/ni/journal/v11/n5/abs/ni.1863.html [2] Holvoet P, Davey PC, Keyzer D De, Doukouré M, Deridder E, Bochaton-Piallat M-L, Gabbiani G, Beaufort E, Bishay K, Andrieux N, Benhabilès N, Marguerie G. Oxidized low-density lipoprotein correlates positively with toll-like receptor 2 and interferon regulatory factor-1 and inversely with superoxide dismutase-1 expression: studies in hypercholesterolemic swine and THP-1 cells. Arterioscler. Thromb. Vasc. Biol. 2006 ;26(7):1558–65. atvb.ahajournals.org/content/26/7/1558 [3] Seimon T a, Nadolski MJ, Liao X, Magallon J, Nguyen M, Feric NT, Koschinsky ML, Harkewicz R, Witztum JL, Tsimikas S, Golenbock D, Moore KJ, Tabas I. Atherogenic lipids and lipoproteins trigger CD36-TLR2-dependent apoptosis in macrophages undergoing endoplasmic reticulum stress. Cell Metab. 2010 ;12(5):467–82. www.cell.com/cell-metabolism/fulltext/S1550-4131(10)00346-3 [4] Seneviratne A, Monaco C. Role Of Inflammatory Cells And Toll-Like Receptors In Atherosclerosis. Curr. Vasc. Pharmacol. 2013 www.eurekaselect.com/116000/article [5] Lang KS, Recher M, Junt T, Navarini AA, Harris NL, Freigang S, Odermatt B, Conrad C, Ittner LM, Bauer S, Luther SA, Uematsu S, Akira S, Hengartner H, Zinkernagel RM. Toll-like receptor engagement converts T-cell autoreactivity into overt autoimmune disease. Nat Med. 2005 ;11(2):138–145. www.nature.com/articles/nm1176 [6] Zhu J, Quyyumi AA, Norman JE, Csako G, Waclawiw MA, Shearer GM, Epstein SE. Effects of total pathogen burden on coronary artery disease risk and C-reactive protein levels. Am. J. Cardiol. 2000 ;85(2):140–6. www.sciencedirect.com/science/article/pii/S0002914999006530 [7] Haidari M, Wyde PR, Litovsky S, Vela D, Ali M, Casscells SW, Madjid M. Influenza virus directly infects, inflames, and resides in the arteries of atherosclerotic and normal mice. Atherosclerosis. 2010 ;208(1):90–96. www.sciencedirect.com/science/article/pii/S0021915009005917 [8] Ibrahim AI, Obeid MT, Jouma MJ, Moasis GA, Al-Richane WL, Kindermann I, Boehm M, Roemer K, Mueller-Lantzsch N, Gärtner BC. Detection of herpes simplex virus, cytomegalovirus and Epstein-Barr virus DNA in atherosclerotic plaques and in unaffected bypass grafts. J. Clin. Virol. 2005 ;32(1):29–32. www.sciencedirect.com/science/article/pii/S1386653204001891 [9] Seneviratne AN, Edsfeldt A, Cole JE, Kassiteridi C, Swart M, Park I, Green P, Khoyratty T, Saliba D, Goddard ME, Sansom SN, Goncalves I, Krams R, Udalova IA, Monaco C. Interferon Regulatory Factor 5 Controls Necrotic Core Formation in Atherosclerotic Lesions by Impairing Efferocytosis. Circulation. 2017 ;136(12):1140–1154. circ.ahajournals.org/content/136/12/1140 [10] Lopez AD, Mathers CD, Ezzati M, Jamison DT, Murray CJ. Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data. 2006 ;367(9524):1747–1757. http://www.thelancet.com/journals/lancet/article/PIIS0140-6736(06)68770-9/fulltext Comments are closed.
|
AuthorDr. Anusha Seneviratne  This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. Categories
All
Archives
March 2020
|