|
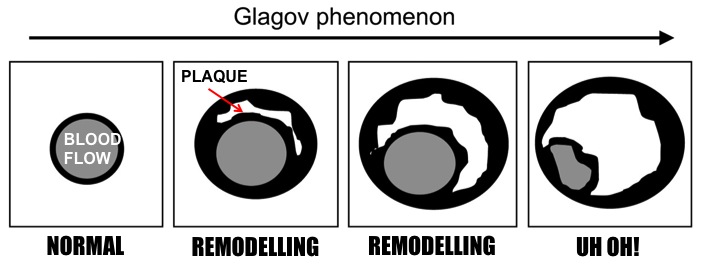
Just six weeks before his death in 2008, American TV journalist Tim Russert underwent a cardiac stress test, as he was already known to have coronary artery disease. He exercised on a treadmill while his blood pressure and heart were monitored. Despite being overweight, hypertensive and having excess cholesterol, which were controlled by medication and lifestyle changes, Russert did not display any symptoms suggestive of imminent cardiac problems. Yet within two months, he died of a sudden cardiac arrest. How did doctors miss this impending cardiac event? It seems that the body’s defence mechanisms eluded existing medical technology. As you may have read before (Blog post: Blocked Pipework in Our Hearts), our arteries detect that a growing atherosclerotic plaque can reduce blood flow to the intended tissues, so the artery wall expands outwards in an attempt to maintain the diameter of the artery through which blood flows. This process is referred to as positive remodelling or ‘Glagov remodelling’, named after the late Professor Seymour Glagov who demonstrated this phenomenon. Standard angiography – where a dye is administered into the patient’s coronary arteries and X-ray imaging is used to visualise arterial blood flow – will only reveal significant blockages or narrowing of arteries. But if an artery has remodelled considerably to maintain blood flow, atherosclerotic plaques will not be detected in spite of how large they may be. In fact, up to 75% of ruptured atherosclerotic plaques, which have significantly reduced or blocked blood flow, exhibit positive remodelling [1]. Once the plaque reaches the ‘Glagovian’ limit of blocking 40% of the cross-sectional area of the artery, expansion can no longer occur so further plaque growth only serves to block blood flow, otherwise referred to as negative remodelling [2]. This is when arterial disease will be detected by angiography. As described previously, smooth muscle cells from the artery wall move into the developing plaque and create a layer on top full of smooth muscle cells and collagen produced by these cells, called the fibrous cap. When this cap becomes extremely thin and weak, due to proteins produced by macrophages (the main inflammatory cell) breaking it down, the cap ruptures and can trigger the appearance of a blood clot. If a blood clot does not appear, the ruptured cap will heal itself with the further production of collagen from the smooth muscle cells. The plaque may undergo several cycles of rupture and repair without any blood clots and subsequent symptoms appearing. And it is this process that determines the speed at which the transition from positive to negative remodelling occurs. If the plaque is permitted to grow with the gradual intrusion of lipids and inflammatory cells, the transition may take decades. However repeating cycles of rupture and repair will rapidly increase the rate at which the plaque grows, thus a seemingly harmless plaque can suddenly present a major threat within months or even weeks. This chain of events was most likely responsible for the sudden and unforeseen demise of Tim Russert. So is there any hope of detecting these rapidly developing plaques before it is too late? Thanks to scientists like Professor Jagat Narula (whose recent lecture at Imperial College is the inspiration behind this blog post), there is now technology available to detect these invisible killers. Optical coherence tomography (OCT) imaging utilising near-infrared light, can be used to detect fibrous caps that are thinner than 65μm (μm = microns or micrometres, one thousandth of a millimetre), which are at high risk of rupturing. Computed tomography (CT) angiograms - where an iodine-rich dye is injected into the patient and then a CT scan of the blood vessels is performed – can be used to observe the necrotic core of plaques (full of dead or dying macrophages and lipids), which is linked to extensive remodelling of the artery, and the presence of calcium deposits in plaques; also indicative of an increased risk of clinical events [1]. This is an extremely important development as a higher degree of remodelling and larger necrotic cores are associated with an earlier onset of acute cardiovascular events. 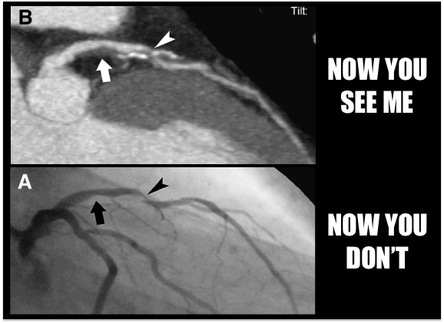 Top: CT image where two plaque are pointed out. The left-hand arrow shows a large plaque, right-hand arrow shows a calcified plaque (calcification in white). Bottom: Angiogram where blood flow appears in grey. Left-hand arrow shows the large plaque in the above image but blood flow is unaffected. Right-hand arrow shows blocked blood flow due to the calcified plaque. Images from Hoffmann et al, (2006) Coronary CT Angiography, J Nucl Med, 47(5): 797-806. It now appears that a combination of CT angiography, positron emission tomography (PET) and the use of FDG (if you really want to know what it stands for, it’s 2-fluorodeoxyglucose) as a contrast agent can be used to visualise plaques with a high level of macrophage-based inflammation [1]. As inflammatory cells have a high rate of metabolism, consuming larger amounts of glucose than other cells to generate energy for their demanding inflammatory activities, FDG acts like glucose and is taken up by these macrophages. And as the FDG is labelled with fluorine-18, a radioisotope emitting positrons as it decays, the PET scanner detects these positrons being emitted at a high rate within the plaque macrophages. Mannose is another sugar taken up by macrophages, and macrophages with the mannose receptor on their cell surface are more prevalent in high-risk plaques. Subsequently fluorine-18 labelled FDM (2-fluorodeoxymannose) has also been successfully used to image macrophage-based inflammation in atherosclerotic plaques [3]. In fact, macrophages take up a greater amount of FDM than FDG, especially when they are starved of oxygen (a condition called hypoxia) in the plaque, which has no direct blood supply until small leaky blood vessels grow into it later on. Thus FDM maybe a better option to image inflammatory plaques. While there is still work to do to improve the quality of these imaging methods and ease the discomfort for patients during the procedure, it is only matter of time until we can identify these dangerous high-risk plaques before it is too late. Atherosclerosis is broadly considered a disease of modern times caused by processed and fatty foods, low levels of exercise or old age. While these causes may apply to today’s patients, it is not a disease exclusive to current generations. CT scans of Egyptian, Peruvian, Native American and Russian mummies have detected calcified atherosclerosis in coronary arteries supplying blood to the heart, and carotid arteries supplying blood to the head, as is commonly seen in patients today [4]. But how could they possibly get atherosclerosis when their diets and lifestyles were completely different? Further examination of the mummies showed these individuals had black lungs, much like smokers of today. Thousands of years ago people relied on wood- or coal-fuelled fire for warmth and cooking, and were thus highly exposed to smoke and soot. Infectious diseases were also more prevalent, and high levels of chronic inflammation caused by infections are known to increase atherosclerosis. So as is commonly advised, don’t eat junk and don’t smoke. Not only will it keep your arteries clean, but do you really want to be the smelliest person on the train? I certainly won’t sit next to you….
References 1. Narula J, Nakano M, Virmani R, Kolodgie FD, Petersen R, Newcomb R, Malik S, Fuster V, Finn A V., Histopathologic characteristics of atherosclerotic coronary disease and implications of the findings for the invasive and noninvasive detection of vulnerable plaques., J. Am. Coll. Cardiol. 61 (10) (2013), 1041–51. 2. Finn A V, Nakano M, Narula J, Kolodgie FD, Virmani R., Concept of vulnerable/unstable plaque., Arterioscler. Thromb. Vasc. Biol. 30 (7) (2010), 1282–92. 3. Tahara N, Mukherjee J, Haas HJ de, Petrov AD, Tawakol A, Haider N, Tahara A, Constantinescu CC, Zhou J, Boersma HH, Imaizumi T, Nakano M, Finn A, Fayad Z, Virmani R, Fuster V, Bosca L, Narula J., 2-deoxy-2-[18F]fluoro-D-mannose positron emission tomography imaging in atherosclerosis., Nat. Med. 20 (2) (2014), 215–9. 4. Thompson RC, Allam AH, Lombardi GP, Wann LS, Sutherland ML, Sutherland JD, Soliman MA-T, Frohlich B, Mininberg DT, Monge JM, Vallodolid CM, Cox SL, Abd el-Maksoud G, Badr I, Miyamoto MI, el-Halim Nur el-Din A, Narula J, Finch CE, Thomas GS., Atherosclerosis across 4000 years of human history: the Horus study of four ancient populations., Lancet 381 (9873) (2013), 1211–22.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorDr. Anusha Seneviratne  This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. Categories
All
Archives
March 2020
|